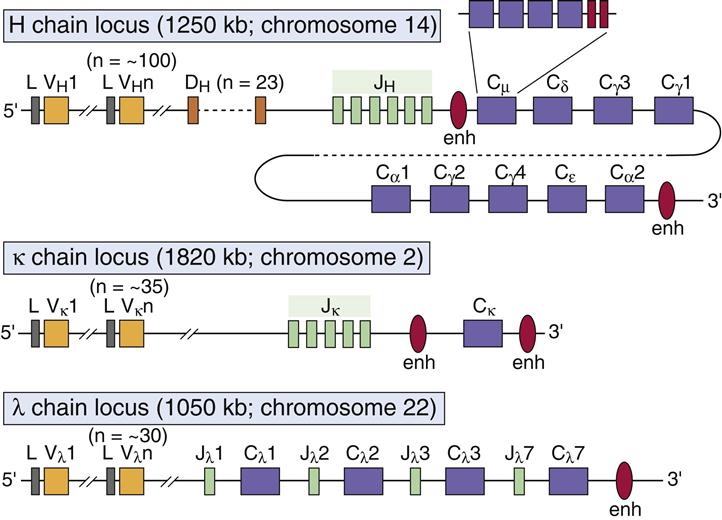
Insertion of constant region domains of human IgG1 into CD4-PE40 increases its plasma half-life. Batra, J.K., Kasturi, S., Gallo, M.G., Voorman, R.L., Maio, S.M., Chaudhary, V.K., Pastan, I.Galactosylation of serum IgG and autoantibodies in murine models of autoimmune haemolytic anaemia. Barker, R.N., Young, R.D., Leader, K.A., Elson, C.J.A monoclonal antibody raised by immunising mice with group A streptococci binds to agalactosyl IgG from rheumatoid arthritis. Rook, G.A., Steele, J., Rademacher, T.The effect of synthetic peptides-corresponding to the amino acid sequences 289-301 (Y48) and 293-301 (Y91) within the CH-2 domain in the human IgG1 was studied on the oxazolone-specific primary and secondary antibody response isotype distribution and on the sheep erythrocyte (SRBC)-specific primary IgM response.An aglycosyl variant of this immunoglobulin, in which site-directed mutagenesis had been used to eliminate the carbohydrate attachment site in the CH2 domain by changing Asn297 to Ala, was compared in this system to aglycosyl immunoglobulin prepared from immunoglobulin-secreting cells treated with tunicamycin to inhibit N-linked glycosylation.For both IgG1 and IgM fusion proteins, the best expression in COS cells was observed for molecules lacking the CH1 domain of the heavy-chain constant region.Gel analysis of specifically immunoprecipitated nascent chains indicates that the carbohydrate moiety can be added to the nascent heavy chains very soon after the presumptive asparaginyl glycosylation site ( CH2 domain) is synthesized on the ribosome.The M3.11 cell line produces an Ig with a polypeptide deletion involving the CH3 domain.Loss of a consensus splice signal in a mutant immunoglobulin gene eliminates the CH1 domain exon from the mRNA.These findings suggest that, during their evolution, a double unequal crossing-over event has taken place at different intervening sequences, resulting in the transfer of the DNA segment coding for the CH1 domain or CH1-CH2 domains.The base sequence upstream from the junction, however, is unrelated to the amino acid sequence of the CH1 domain and the hinge region of all the gamma chains whose sequences have been determined.The serum half-lives of a wild-type recombinant mouse monoclonal antibody of the IgG2b isotype and a mutant antibody differing from the wild-type antibody by a single amino acid substitution introduced into the CH2 domain, the replacement of Asn 297 by Ala to delete the conserved site of heavy chain glycosylation, were determined in the rat.These mice possess two CH2 IgH-1b and two CH2 Igh-1a/CH2 Igh-1d determinants, suggesting that these variant immunoglobulin arose from recombinations within the CH2 domain.(2) Among the eight anti-IgA subclass antibodies, seven were directed against the CH2 domain of IgA whereas the anti-IgA1 1-155-1 recognised an epitope destroyed by Streptococcus sanguis IgA1 protease and localised in the hinge region of IgA1.Glycosylated chimeric mouse-human anti-NIP IgG3 antibody produced by growth of the J558L mouse B cell plasmacytoma is characterised with respect to the single carbohydrate chain at Asn-297 in the CH2 domain indicating that the mouse cell glycosyl transferases dictate the pattern of glycosylation rather than the human CH region of the heavy chain.CD4-PE40 is a recombinant toxin containing the binding domain of CD4 and a mutant form of Pseudomonas exotoxin A from which the cell binding domain has been removed.A number of systemic autoimmune diseases are associated with increased levels of the agalactosyl (G0) IgG isoforms that lack a terminal galactose from the CH2 domain oligosaccharide.It was shown recently that the IgG of patients with rheumatoid arthritis tends to lack the terminal galactose normally present on the conserved N-linked oligosaccharide situated on the CH2 domain.
